Analysis

A Promising Outlook for Digital Therapeutics (DTx) in Europe – how can biopharma capitalise?
The Digital Therapeutics (DTx or PDTs) market has taken large strides in the last few years, growing from infancy to a mature, well-respected older version of its predecessor, with widely developing business models and use-cases available to participate in commercial activities alongside other, more traditional pharmaceutical players. Regulators around the globe are also developing frameworks for research & development, clinical validation, as well as the general regulatory requirements needed for entering the market. 1, 2
Driven by technological developments, healthcare cost pressure and consumer demand for convenience, the Digital Therapeutics (DTx, see below for a detailed overview) funding, has grown significantly over the past few years3 . Among the three continents – Asia, America and Europe, Europe is currently uniquely positioned to be a leader for DTx clear market access pathways into the public health systems with regulations currently existing in Germany, France, Belgium and recently Austria). Thanks to the regulatory pathways’ development in Europe, the above-mentioned EU markets often acts as an attractive market entry point, even for non-EU based companies.
In recent years, a lot has changed in terms of DTx regulations, adoption, and usage in Europe. In this report, we not only describe the status quo of DTx in Europe, but we also investigate – what we believe – is a promising future for DTx in Europe and what are best practices for DTx and biopharma companies in creating successful business models in DTx.
DTx growing market brings new opportunities to life-science companies (Biopharma and MedTech). DTx offer industry stakeholders an opportunity to craft more personalised experiences and new ways to become patient-centric.
Digital therapeutics offer industry stakeholders an opportunity to craft more patient-centric approaches
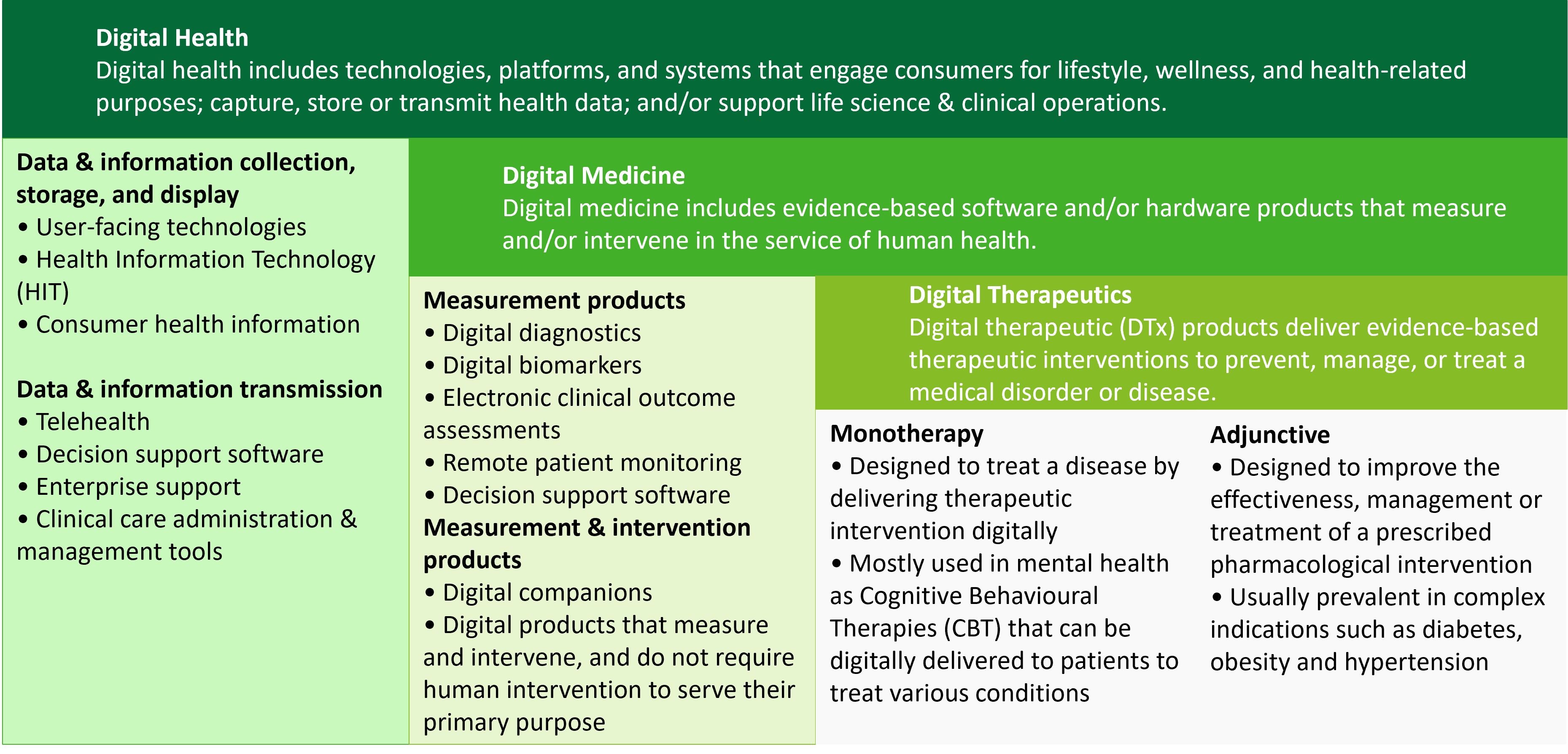
DTx are patient-facing software applications designed to help patients treat, prevent, monitor and effectively self-manage a disease and those that have a proven clinical benefit. These solutions can function as standalone therapies or be prescribed in combination with medication, often leading to improved patient adherence. Digital health, digital medicine, and digital therapeutics also offer industry stakeholders an opportunity to craft more personalised experiences and new ways to become patient-centric.
Differences between digital health, digital medicine and digital therapeutics
Reasons for optimism – How could life-science companies benefit from adopting DTx strategy into their portfolio?
Even in difficult times, some important factors may still make DTx an above-average attractive segment for investors:
- The Covid-19 pandemic accelerated the confluence of many trends in the healthcare sector, most notably consumers' preference for accessibility and convenience.
- Based on experience from the past three years, both DTx innovators and investors now can develop much more robust business plans and investment cases.
- More DTx companies move from no/single approved product companies to platform/portfolio of products companies.
- The reimbursement levels for DTx vary, but these are on average around 250 to 300 EUR per therapy (usually 6 weeks) in Germany.
What should life-science companies consider when developing DTx?

Biopharma companies need to assess their own capabilities very carefully and must acknowledge that DTx development requires a significantly different skillsets compared to drug development. Major considerations include the time and cost to build internal capabilities, which includes attracting the right talent vs. compromising on control regarding product features, user experience, ownership of data when partnering with a DTx developer.
State of DTx in Europe
In Europe, 2019 marked a significant milestone for digital therapeutics as they gained traction, with Germany pioneering the first regulatory pathway for DTx in Europe with many European countries following in its steps. DTx legislation is currently fragmented among European countries, therefore it is important to choose the best market access points. National regulatory differences lead to variations in which documents are submitted, which in return affects the amount of time spent preparing such documents. This has a direct impact on the evaluation time and key decisions, such as whether to follow fast-track versus standard procedures, reimbursement rates and even the likelihood of receiving reimbursement for the digital solution at all.
Germany
Germany is generally recognised as a DTx reimbursement framework pioneer with an active fast-track model, which has been an inspiration for other European countries. The Digital Healthcare Act (DVG) waspassed in December 2019, and it stipulates reimbursement criteria for patient-oriented digital health applications (DiGAs) that are Medical Devices of low risk (must be classified as a class I or IIa medical device according to Regulation (EU) 2017/745 on medical devices or according to Directive 93/42/EEC).
France
French president recently declared that there would be increased focus on digital health solutions and intention to learn from experiences and mistakes made during the first few years of DiGA in Germany. ThePrise en Charge Anticipée “PEC-AN”, which includes a fast track component, has been implemented as a result.
Belgium
In late January 2021, the National Institute for Health and Disability Insurance (NIHDI) announced that the Insurance Committee had given the go-ahead for medical reimbursement of medical applications (mobile health applications or mHealth applications). Any manufacturers with ambitions to have digital therapeutics approved for reimbursement which goes through an approval process to reach the top of Belgium’s mHealth “pyramid”, can now get their app reimbursed by NIHDI.
There are many countries in Europe that are trying to learn from the experiences of the pioneers, in order adjust and customise and create a local solution.
United Kingdom
The UK is one of the other few countries with a somewhat standardised pathway for DTx (called digital health technology, or DHT in the UK legislation) assessment and financing through Public Insurance Coverage. However, a robust and direct access to statutory reimbursement is still missing.
Spain
In accordance with the ongoing trends of the healthcare sector which focus heavily on patient-centricity and value-based solutions, the digitalisation of the industry has been recognised as a main priority in Spain. As the evaluation process is strictly decentralised, good selection of the region in Spain is crucial for future success.
Austria
In Chapter 7.4 of the recently released Digital Austria Act (DAA), the government parties are proposing the introduction of quality approved DiGA.
Summary, outlook, and invitation
In the short-term, increased complexity is to be anticipated as more countries continue to adopt regulatory frameworks for DTx. However, in the long-term, an EU-wide harmonisation could significantly accelerate DTx business models by cutting down on time-to-market and development cost by avoiding duplication.
For broader perspective and more information please reach out to our full study on the opportunities of DTx in Europe.
_____________________________
1Guidances with Digital Health Content | FDA (2023); URL: https://www.fda.gov/medical-devices/digital-health-center-excellence/guidances-digital-health-content; retrieved on: 25. 10. 2023.
2BfArM - Digital Health Applications (DiGA); URL:
https://www.bfarm.de/EN/Medical-devices/Tasks/DiGA-and-DiPA/Digital-Health-Applications/_node.html;
retrieved on: 25. 10. 2023.
3Digital Therapeutics – medical intervention
beyond the pill (2022); URL: https://dealroom.co/blog/digital-therapeutics-startups-report-2022; retrieved on 22. 10. 2023.
4Life Sciences Regulation in Belgium: Overview (2023);
URL: Life Sciences Regulation in Belgium: Overview |
Practical Law (thomsonreuters.com); retrieved on: 4. 10. 2023.
5mHealth Belgium; URL: Belgian
platform for medical mobile applications - mHealthBELGIUM; retrieved on: 4. 10. 2023.
6Pricing and Reimbursement Laws 2023/ Belgium (2023); URL:
Pricing & Reimbursement Laws and Regulations |
Belgium | GLI (globallegalinsights.com); retrieved on: 4. 10. 2023.
7Price en chargé anticipée numerique (PECAN) (2023); URL: Prise en charge anticipée numérique (PECAN) | G_NIUS
(esante.gouv.fr); retrieved on: 6. 10. 2023.
8Reimbursement profiles; URL: Reimbursement profiles | G_NIUS (esante.gouv.fr); retrieved on: 6. 10. 2023.
9Improving access to digital therapeutics in
Europe (2023); URL: improving-access-to-digital-therapeutics-in-europe.pdf
(efpia.eu); retrieved on: 6. 10. 2023.
10Digital therapeutics alliance; URL: Home
- Digital Therapeutics Alliance (dtxalliance.org),; retrieved on: 6. 10. 2023.
11Evidence standard framework for (ESF) for
digital health companies; URL: Evidence standards framework (ESF) for digital health technologies | Our programmes | What we do | About | NICE, retrieved on: 6. 10. 2023.
12Market access pathways for digital health
solutions (November 2020); URL: 20062_COCIR_Market_Access_Pathways_Digital_Health.pdf; retrieved on: 8. 10. 2023.