Clinical Trial Regulation preparedness has been saved

Solution
Clinical Trial Regulation preparedness
Optimisation of EU CTR readiness for future-proof clinical trials
The EU Clinical Trial Regulation (EU CTR) introduced the Clinical Trial Information System (CTIS) that provides a collaborative platform for sponsors and regulators to manage clinical trials conducted in EU Member States and EEA countries. It promotes patient safety, supports cross-border trials and increases transparency. Sponsors are required to undertake complex organisational transformation to ensure compliance with the EU CTR.
Your challenge
With the go-live of the CTIS on January 31st 2022, and the associated start of the 3-year transition period, clinical trial sponsors have been presented with several tangible risk and challenges regarding conduct of clinical trials in Europe.
- Organisation structure: Geographic presence and representation of organisations where clinical trials are being conducted affects the ability to comply with the EU CTR.
- User administration: Sponsors are required to manage access and rights of all users (internal and contracted research organisation) to all clinical trials across the EU.
- Submission strategy: Parallel submissions are not allowed within the CTIS; therefore, co-ordination and timely document and data readiness across the European landscape is essential.
- Timelines: The EU CTR postulates stricter timelines for sponsors to respond to the request for information that can occur at any point during the validation and assessment, as well as short timelines for notifications on clinical trial milestones and certain events.
- Transparency: Information stored within the CTIS is made publicly available; sponsors must redact all documentation of personal and commercially confidential information.
- Other related regulations: It is imperative to look wider than the EU CTR when considering compliance; IDMP, for example, will play an increasing role for clinical trial master data.
Why Deloitte
Deloitte are uniquely positioned to help organisations optimise and execute EU CTR readiness strategies. While we were not involved in the technical implementation, for the first 5 years of its development we aided EMA in the collation of business requirements and use cases as well as industry and the Member States in the User Acceptance Testing of the CTIS. Since that time, we have also helped several large pharmaceutical companies prepare for EU CTR compliance by defining, executing and improving strategies for EU CTR implementation and clinical trial management through the CTIS.
- We have an enthusiastic and driven team of clinical experts with recent experience of supporting both pharmaceutical organisation and national competent authorities on EU CTR/CTIS compliance and optimisation.
- We have the capabilities to assist compliance and clinical operations by adding accelerators from Regulatory Information Management (RIM) and Master Data Management (MDM) to Operational Services.
- We have and continue to support EMA since 2014 with business services across the majority of their task forces and divisions.
Our solution
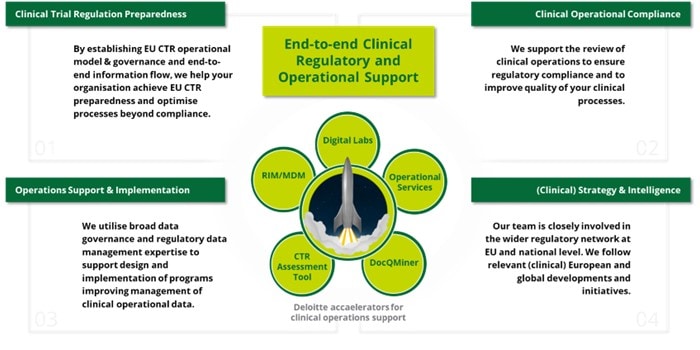
We pride ourselves in providing organisations with end-to-end clinical regulatory and operational support and ensuring they are and remain responsible businesses. We do this by leading and/or supporting implementation, communication and training activities at all levels including programme & project management and subject matter expertise.
- Clinical Trial Regulation Preparedness
We can assist your organisation in achieving EU CTR readiness and improving processes beyond compliance by implementing an operational model and governance for EU CTR, as well as ensuring end-to-end information flow. Our services include offering essential factors to consider when transitioning trials, as well as guiding sponsors through the necessary steps to develop an optimal strategy for transitioning applications/trials to the EU CTR. Overall, we help your organisation take the right steps from foundational EU CTR readiness to an optimised clinical strategy.
- Clinical Operational Compliance
Our services focus on supporting the review of clinical operations to ensure regulatory compliance and improve the quality of clinical processes. We provide compliance assurance in line with ICH E6 R2, EudraLex Volume 10, and EMA guidelines, including our considerations as they relate to EMA’s Reflection paper on the use of Artificial Intelligence (AI) in the medicinal product lifecycle, as well as implementing quality risk management throughout the supply chain. Additionally, we conduct analysis of effective usage of contract research organisations and contract manufacturing organisations (CROs and CMOs).
- Operations Support & Implementation
Our extensive knowledge in data governance and regulatory data management allows us to assist in designing and implementing programmes that improve the management of clinical operational data. Our services include conducting assessments and developing strategies for clinical operational data management, providing independent software vendor selection support, and assisting with CTMS implementation and training.
- (Clinical) Strategy & Intelligence
Our team of clinical experts is interconnected with the wider clinical network by following closely current developments and initiatives like ACT EU (Accelerating Clinical Trials in the EU), DARWIN EU (Data Analysis and Real World Interrogation Network) and ICH M11 CeSHarP (Clinical electronic Structured Harmonized Protocol).
Recommendations
EU Clinical Trial Regulation & Clinical Trial Information System
Optimization EU CTR Readiness